
While lithium-ion batteries offer several advantages over lead-acid batteries, they also have some disadvantages: Top 5 disadvantages of lithium-ion batteries This implies that lithium-ion batteries can store more energy per unit of weight and volume, making them more suitable for portable and lightweight applications. 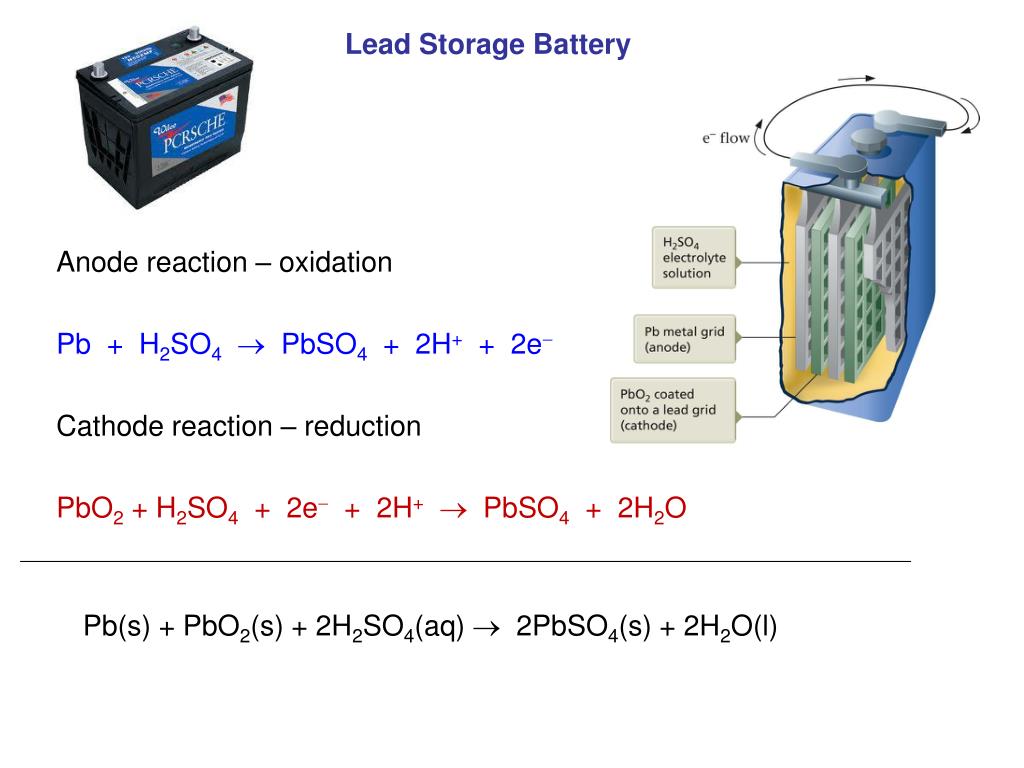
Lead-acid batteries have a lower energy density (30-50 Wh/kg) and specific energy (20-50 Wh/L) compared to lithium-ion batteries (150-200 Wh/kg and 250-670 Wh/L, respectively). The fundamental electrochemical process in these batteries involves the reversible intercalation and deintercalation of lithium ions between the anode and cathode materials. In contrast, lithium-ion batteries consist of an anode typically made of graphite, a cathode made of lithium metal oxide (such as LiCoO2, LiFePO4, or LiNiMnCoO2), and an electrolyte composed of lithium salts dissolved in a solvent (usually a mixture of organic carbonates). Chemistry and Components Lithium-ion batteries The electrochemical reactions within the cells involve the conversion of lead and lead dioxide to lead sulfate (PbSO4) upon discharging, and the reverse process during charging. The lead-acid battery comprises a set of electrochemical cells, each consisting of a lead (Pb) anode, a lead dioxide (PbO2) cathode, and a sulfuric acid (H2SO4) electrolyte solution. Chemistry and Components Lead-acid batteries 
Both types of batteries serve as power storage devices with distinct advantages and disadvantages, depending on the application. As technical engineers specializing in the design of lead-acid battery recycling plants, GME’s team would like to provide a detailed and informative comparison between lead-acid and lithium-ion batteries.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
